You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000000147_01963
You are here: Home > Sequence: MGYG000000147_01963
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
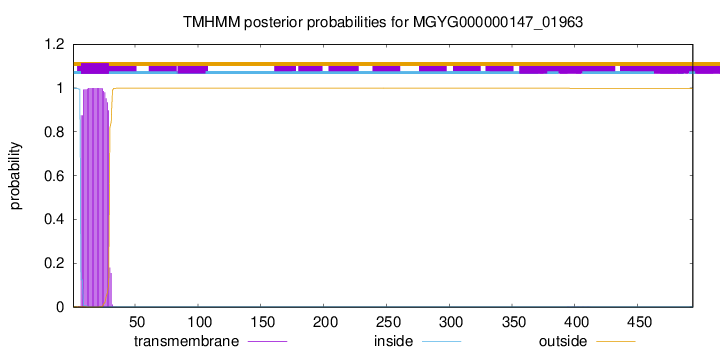
TMHMM annotations
Basic Information help
| Species | Bacillus paralicheniformis | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Firmicutes; Bacilli; Bacillales; Bacillaceae; Bacillus; Bacillus paralicheniformis | |||||||||||
| CAZyme ID | MGYG000000147_01963 | |||||||||||
| CAZy Family | PL1 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 177863; End: 179347 Strand: - | |||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| PL1 | 223 | 417 | 2.5e-95 | 0.9948717948717949 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| COG3866 | PelB | 5.12e-71 | 162 | 478 | 17 | 341 | Pectate lyase [Carbohydrate transport and metabolism]. |
| smart00656 | Amb_all | 1.29e-39 | 228 | 417 | 3 | 187 | Amb_all domain. |
| pfam00544 | Pec_lyase_C | 1.87e-36 | 211 | 416 | 8 | 211 | Pectate lyase. This enzyme forms a right handed beta helix structure. Pectate lyase is an enzyme involved in the maceration and soft rotting of plant tissue. |
| pfam14200 | RicinB_lectin_2 | 0.006 | 73 | 169 | 4 | 89 | Ricin-type beta-trefoil lectin domain-like. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| BCE07889.1 | 0.0 | 1 | 494 | 1 | 494 |
| BCE15879.1 | 0.0 | 1 | 494 | 1 | 494 |
| BCE09706.1 | 0.0 | 1 | 494 | 1 | 494 |
| BAL45990.1 | 0.0 | 1 | 494 | 1 | 494 |
| AJO19478.1 | 0.0 | 1 | 494 | 1 | 494 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 3ZSC_A | 1.05e-12 | 181 | 393 | 8 | 212 | Catalyticfunction and substrate recognition of the pectate lyase from Thermotoga maritima [Thermotoga maritima] |
| 5AMV_A | 1.93e-10 | 232 | 393 | 118 | 294 | Structuralinsights into the loss of catalytic competence in pectate lyase at low pH [Bacillus subtilis],5X2I_A Polygalacturonate Lyase by Fusing with a Self-assembling Amphipathic Peptide [Bacillus subtilis subsp. subtilis str. 168] |
| 1BN8_A | 2.08e-10 | 232 | 393 | 139 | 315 | BacillusSubtilis Pectate Lyase [Bacillus subtilis] |
| 3KRG_A | 4.53e-10 | 232 | 393 | 118 | 294 | ChainA, Pectate lyase [Bacillus subtilis] |
| 2BSP_A | 4.86e-10 | 232 | 393 | 139 | 315 | ChainA, PROTEIN (PECTATE LYASE) [Bacillus subtilis] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| O34819 | 5.84e-127 | 164 | 483 | 24 | 341 | Pectin lyase OS=Bacillus subtilis (strain 168) OX=224308 GN=pelB PE=3 SV=1 |
| P94449 | 6.66e-126 | 164 | 483 | 24 | 341 | Pectin lyase OS=Bacillus subtilis OX=1423 GN=pelB PE=1 SV=1 |
| P27027 | 2.06e-102 | 175 | 487 | 10 | 312 | Pectin lyase OS=Pseudomonas marginalis OX=298 GN=pnl PE=1 SV=2 |
| P24112 | 2.28e-93 | 172 | 483 | 4 | 310 | Pectin lyase OS=Pectobacterium carotovorum OX=554 GN=pnl PE=1 SV=1 |
| Q00645 | 7.64e-19 | 190 | 393 | 43 | 234 | Pectate lyase plyB OS=Emericella nidulans (strain FGSC A4 / ATCC 38163 / CBS 112.46 / NRRL 194 / M139) OX=227321 GN=plyB PE=1 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as SP
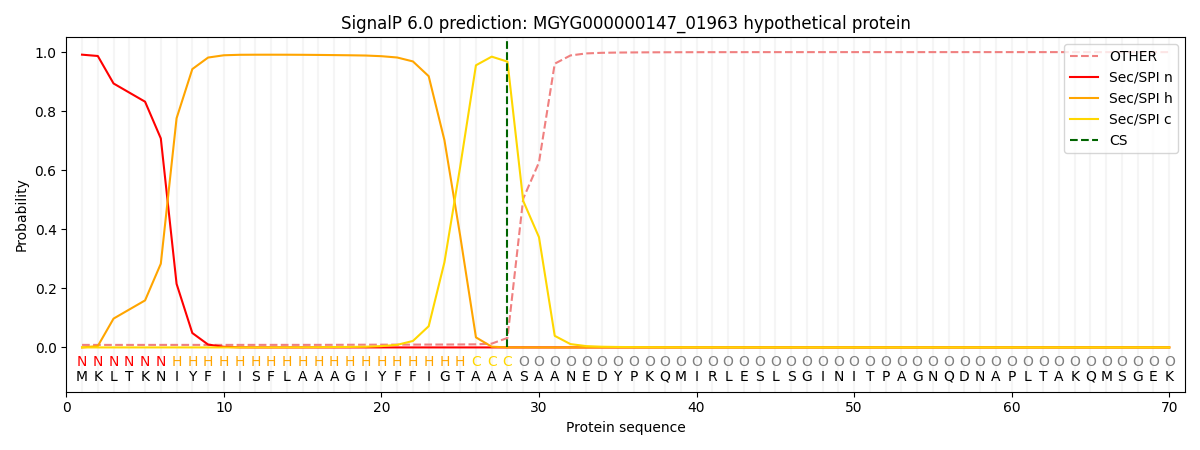
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.010921 | 0.987893 | 0.000407 | 0.000269 | 0.000231 | 0.000243 |