You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000001690_00156
You are here: Home > Sequence: MGYG000001690_00156
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Ruminococcus_G gauvreauii | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Firmicutes_A; Clostridia; Lachnospirales; Lachnospiraceae; Ruminococcus_G; Ruminococcus_G gauvreauii | |||||||||||
| CAZyme ID | MGYG000001690_00156 | |||||||||||
| CAZy Family | CE4 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 174983; End: 176482 Strand: + | |||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| CE4 | 303 | 413 | 4.1e-25 | 0.8307692307692308 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| cd10954 | CE4_CtAXE_like | 9.62e-82 | 304 | 481 | 3 | 180 | Catalytic NodB homology domain of Clostridium thermocellum acetylxylan esterase and its bacterial homologs. This family is represented by Clostridium thermocellum acetylxylan esterase (CtAXE, EC 3.1.1.72), a member of the carbohydrate esterase 4 (CE4) superfamily. CtAXE deacetylates O-acetylated xylan, a key component of plant cell walls. It shows no detectable activity on generic esterase substrates including para-nitrophenyl acetate. It is specific for sugar-based substrates and will precipitate acetylxylan, as a consequence of deacetylation. CtAXE is a monomeric protein containing a catalytic NodB homology domain with the same overall topology and a deformed (beta/alpha)8 barrel fold as other CE4 esterases. However, due to differences in the topography of the substrate-binding groove, the chemistry of the active center, and metal ion coordination, CtAXE has different metal ion preference and lacks activity on N-acetyl substrates. It is significantly activated by Co2+. Moreover, CtAXE displays distinctly different ligand coordination to the metal ion, utilizing an aspartate, a histidine, and four water molecules, as opposed to the conserved His-His-Asp zinc-binding triad of other CE4 esterases. |
| cd10917 | CE4_NodB_like_6s_7s | 3.46e-61 | 303 | 469 | 2 | 171 | Catalytic NodB homology domain of rhizobial NodB-like proteins. This family belongs to the large and functionally diverse carbohydrate esterase 4 (CE4) superfamily, whose members show strong sequence similarity with some variability due to their distinct carbohydrate substrates. It includes many rhizobial NodB chitooligosaccharide N-deacetylase (EC 3.5.1.-)-like proteins, mainly from bacteria and eukaryotes, such as chitin deacetylases (EC 3.5.1.41), bacterial peptidoglycan N-acetylglucosamine deacetylases (EC 3.5.1.-), and acetylxylan esterases (EC 3.1.1.72), which catalyze the N- or O-deacetylation of substrates such as acetylated chitin, peptidoglycan, and acetylated xylan. All members of this family contain a catalytic NodB homology domain with the same overall topology and a deformed (beta/alpha)8 barrel fold with 6- or 7 strands. Their catalytic activity is dependent on the presence of a divalent cation, preferably cobalt or zinc, and they employ a conserved His-His-Asp zinc-binding triad closely associated with the conserved catalytic base (aspartic acid) and acid (histidine) to carry out acid/base catalysis. Several family members show diversity both in metal ion specificities and in the residues that coordinate the metal. |
| cd10947 | CE4_SpPgdA_BsYjeA_like | 1.66e-59 | 304 | 478 | 3 | 177 | Catalytic NodB homology domain of Streptococcus pneumoniae peptidoglycan deacetylase PgdA, Bacillus subtilis BsYjeA protein, and their bacterial homologs. This family is represented by Streptococcus pneumoniae peptidoglycan GlcNAc deacetylase (SpPgdA), a member of the carbohydrate esterase 4 (CE4) superfamily. SpPgdA protects gram-positive bacterial cell wall from host lysozymes by deacetylating peptidoglycan N-acetylglucosamine (GlcNAc) residues. It consists of three separate domains: N-terminal, middle and C-terminal (catalytic) domains. The catalytic NodB homology domain is similar to the deformed (beta/alpha)8 barrel fold adopted by other CE4 esterases, which harbors a mononuclear metalloenzyme employing a conserved His-His-Asp zinc-binding triad closely associated with conserved catalytic base (aspartic acid) and acid (histidine) to carry out acid/base catalysis. The enzyme is able to accept GlcNAc3 as a substrate, with the N-acetyl of the middle sugar being removed by the enzyme. This family also includes Bacillus subtilis BsYjeA protein encoded by the yjeA gene, which is one of the six polysaccharide deacetylase gene homologs (pdaA, pdaB/ybaN, yheN, yjeA, yxkH and ylxY) in the Bacillus subtilis genome. Although homology comparison shows that the BsYjeA protein contains a polysaccharide deacetylase domain, and was predicted to be a membrane-bound xylanase or a membrane-bound chitooligosaccharide deacetylase, more recent research indicates BsYjeA might be a novel non-specific secretory endonuclease which creates random nicks progressively on the two strands of dsDNA, resulting in highly distinguishable intermediates/products very different in chemical and physical compositions over time. In addition, BsYjeA shares several enzymatic properties with the well-understood DNase I endonuclease. Both enzymes are active on ssDNA and dsDNA, both generate random nicks, and both require Mg2+ or Mn2+ for hydrolytic activity. |
| NF033930 | pneumo_PspA | 2.65e-59 | 72 | 281 | 442 | 652 | pneumococcal surface protein A. The pneumococcal surface protein proteins, found in Streptococcus pneumoniae, are repetitive, with patterns of localized high sequence identity across pairs of proteins given different specific names that recombination may be presumed. This protein, PspA, has an N-terminal region that lacks a cross-wall-targeting YSIRK type extended signal peptide, in contrast to the closely related choline-binding protein CbpA which has a similar C-terminus but a YSIRK-containing region at the N-terminus. |
| NF033838 | PspC_subgroup_1 | 2.66e-53 | 92 | 278 | 485 | 672 | pneumococcal surface protein PspC, choline-binding form. The pneumococcal surface protein PspC, as described in Streptococcus pneumoniae, is a repetitive and highly variable protein, recognized by a conserved N-terminal domain and also by genomic location. This form, subgroup 1, has variable numbers of a choline-binding repeat in the C-terminal region, and is also known as choline-binding protein A. The other form, subgroup 2, is anchored covalently after cleavage by sortase at a C-terminal LPXTG site. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| QQQ92356.1 | 5.15e-58 | 295 | 499 | 337 | 542 |
| QJU15090.1 | 5.15e-58 | 295 | 499 | 337 | 542 |
| ANU74806.1 | 7.49e-58 | 295 | 499 | 356 | 561 |
| ASU27615.1 | 7.49e-58 | 295 | 499 | 356 | 561 |
| ASM70448.1 | 1.02e-57 | 304 | 498 | 182 | 375 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 5LFZ_A | 8.85e-47 | 293 | 495 | 16 | 215 | T48deacetylase [Arthrobacter sp. AW19M34-1],5LGC_A T48 deacetylase with substrate [Arthrobacter sp. AW19M34-1] |
| 6H8L_A | 1.08e-39 | 304 | 481 | 12 | 189 | Structureof peptidoglycan deacetylase PdaC from Bacillus subtilis [Bacillus subtilis subsp. subtilis str. 168],6H8L_B Structure of peptidoglycan deacetylase PdaC from Bacillus subtilis [Bacillus subtilis subsp. subtilis str. 168] |
| 6H8N_A | 7.86e-39 | 304 | 481 | 12 | 189 | Structureof peptidoglycan deacetylase PdaC from Bacillus subtilis - mutant D285S [Bacillus subtilis subsp. subtilis str. 168],6H8N_B Structure of peptidoglycan deacetylase PdaC from Bacillus subtilis - mutant D285S [Bacillus subtilis subsp. subtilis str. 168] |
| 2C1G_A | 5.22e-32 | 304 | 481 | 238 | 415 | Structureof Streptococcus pneumoniae peptidoglycan deacetylase (SpPgdA) [Streptococcus pneumoniae R6] |
| 2C1I_A | 2.49e-31 | 304 | 481 | 238 | 415 | Structureof Streptococcus pneumoniae peptidoglycan deacetylase (SpPgdA) D 275 N Mutant. [Streptococcus pneumoniae R6] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| O34798 | 1.60e-36 | 304 | 481 | 280 | 457 | Peptidoglycan-N-acetylmuramic acid deacetylase PdaC OS=Bacillus subtilis (strain 168) OX=224308 GN=pdaC PE=1 SV=1 |
| Q8Y9V5 | 5.16e-35 | 293 | 495 | 257 | 456 | Peptidoglycan-N-acetylglucosamine deacetylase PgdA OS=Listeria monocytogenes serovar 1/2a (strain ATCC BAA-679 / EGD-e) OX=169963 GN=pgdA PE=1 SV=1 |
| A0A0H3GDH9 | 5.16e-35 | 293 | 495 | 257 | 456 | Peptidoglycan-N-acetylglucosamine deacetylase PgdA OS=Listeria monocytogenes serotype 1/2a (strain 10403S) OX=393133 GN=pgdA PE=2 SV=1 |
| A0A3Q0NBH7 | 5.16e-35 | 293 | 495 | 257 | 456 | Peptidoglycan-N-acetylglucosamine deacetylase PgdA OS=Listeria monocytogenes serotype 1/2a (strain EGD / Mackaness) OX=1334565 GN=pgdA PE=1 SV=1 |
| Q8DP63 | 4.40e-31 | 304 | 481 | 270 | 447 | Peptidoglycan-N-acetylglucosamine deacetylase OS=Streptococcus pneumoniae (strain ATCC BAA-255 / R6) OX=171101 GN=pgdA PE=1 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as OTHER
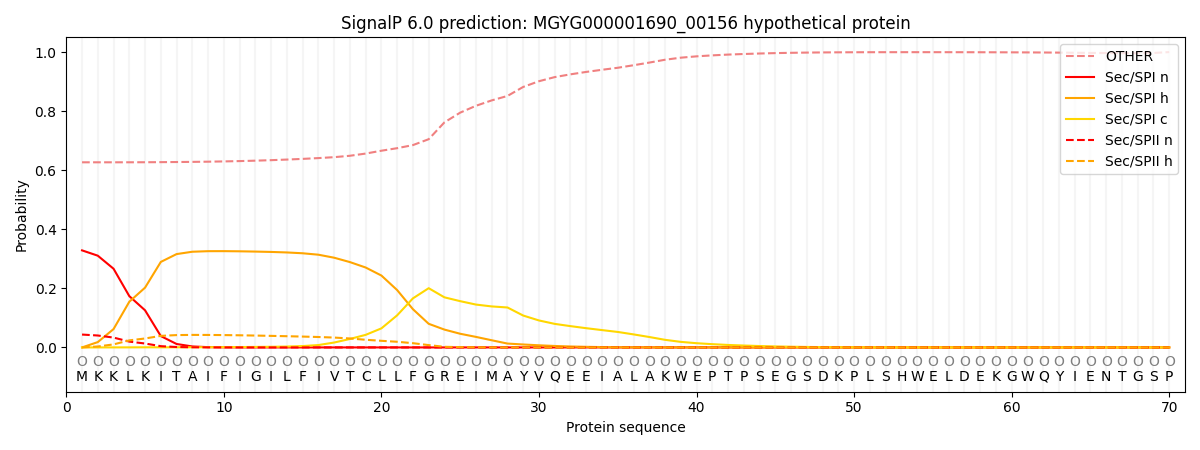
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.640291 | 0.311463 | 0.045300 | 0.000695 | 0.000441 | 0.001804 |

