You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000002300_00749
You are here: Home > Sequence: MGYG000002300_00749
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Bacteroides cutis | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Bacteroidota; Bacteroidia; Bacteroidales; Bacteroidaceae; Bacteroides; Bacteroides cutis | |||||||||||
| CAZyme ID | MGYG000002300_00749 | |||||||||||
| CAZy Family | CE7 | |||||||||||
| CAZyme Description | Acetyl esterase Axe7A | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 950439; End: 951728 Strand: + | |||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| CE7 | 125 | 422 | 3e-85 | 0.9680511182108626 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| COG3458 | Axe1 | 1.15e-40 | 125 | 427 | 9 | 320 | Cephalosporin-C deacetylase or related acetyl esterase [Secondary metabolites biosynthesis, transport and catabolism]. |
| pfam05448 | AXE1 | 2.70e-36 | 125 | 423 | 8 | 316 | Acetyl xylan esterase (AXE1). This family consists of several bacterial acetyl xylan esterase proteins. Acetyl xylan esterases are enzymes that hydrolyze the ester linkages of the acetyl groups in position 2 and/or 3 of the xylose moieties of natural acetylated xylan from hardwood. These enzymes are one of the accessory enzymes which are part of the xylanolytic system, together with xylanases, beta-xylosidases, alpha-arabinofuranosidases and methylglucuronidases; these are all required for the complete hydrolysis of xylan. |
| COG1506 | DAP2 | 1.32e-04 | 173 | 426 | 368 | 618 | Dipeptidyl aminopeptidase/acylaminoacyl peptidase [Amino acid transport and metabolism]. |
| COG0412 | DLH | 1.54e-04 | 189 | 426 | 6 | 235 | Dienelactone hydrolase [Secondary metabolites biosynthesis, transport and catabolism]. |
| COG4287 | PqaA | 0.001 | 269 | 385 | 214 | 347 | PhoPQ-activated pathogenicity-related protein [General function prediction only]. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| ADD61405.1 | 8.17e-285 | 2 | 427 | 1 | 425 |
| QUU00600.1 | 8.17e-285 | 2 | 427 | 1 | 425 |
| QUT62080.1 | 8.17e-285 | 2 | 427 | 1 | 425 |
| ADD62010.1 | 8.17e-285 | 2 | 427 | 1 | 425 |
| QQA30546.1 | 8.17e-285 | 2 | 427 | 1 | 425 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 6AGQ_A | 6.28e-25 | 125 | 407 | 12 | 303 | Acetylxylan esterase from Paenibacillus sp. R4 [Paenibacillus sp. R4],6AGQ_B Acetyl xylan esterase from Paenibacillus sp. R4 [Paenibacillus sp. R4],6AGQ_C Acetyl xylan esterase from Paenibacillus sp. R4 [Paenibacillus sp. R4],6AGQ_D Acetyl xylan esterase from Paenibacillus sp. R4 [Paenibacillus sp. R4],6AGQ_E Acetyl xylan esterase from Paenibacillus sp. R4 [Paenibacillus sp. R4],6AGQ_F Acetyl xylan esterase from Paenibacillus sp. R4 [Paenibacillus sp. R4] |
| 1ODS_A | 1.54e-24 | 125 | 407 | 9 | 298 | CephalosporinC deacetylase from Bacillus subtilis [Bacillus subtilis],1ODS_B Cephalosporin C deacetylase from Bacillus subtilis [Bacillus subtilis],1ODS_C Cephalosporin C deacetylase from Bacillus subtilis [Bacillus subtilis],1ODS_D Cephalosporin C deacetylase from Bacillus subtilis [Bacillus subtilis],1ODS_E Cephalosporin C deacetylase from Bacillus subtilis [Bacillus subtilis],1ODS_F Cephalosporin C deacetylase from Bacillus subtilis [Bacillus subtilis],1ODS_G Cephalosporin C deacetylase from Bacillus subtilis [Bacillus subtilis],1ODS_H Cephalosporin C deacetylase from Bacillus subtilis [Bacillus subtilis] |
| 1L7A_A | 2.11e-24 | 125 | 407 | 9 | 298 | structuralGenomics, crystal structure of Cephalosporin C deacetylase [Bacillus subtilis],1L7A_B structural Genomics, crystal structure of Cephalosporin C deacetylase [Bacillus subtilis] |
| 5GMA_A | 3.89e-24 | 122 | 407 | 26 | 315 | Crystalstructure of the P228A variant of Thermotoga maritima acetyl esterase [Thermotoga maritima MSB8],5GMA_B Crystal structure of the P228A variant of Thermotoga maritima acetyl esterase [Thermotoga maritima MSB8],5GMA_C Crystal structure of the P228A variant of Thermotoga maritima acetyl esterase [Thermotoga maritima MSB8],5GMA_D Crystal structure of the P228A variant of Thermotoga maritima acetyl esterase [Thermotoga maritima MSB8],5GMA_E Crystal structure of the P228A variant of Thermotoga maritima acetyl esterase [Thermotoga maritima MSB8],5GMA_F Crystal structure of the P228A variant of Thermotoga maritima acetyl esterase [Thermotoga maritima MSB8] |
| 1ODT_C | 3.95e-24 | 125 | 407 | 9 | 298 | cephalosporinC deacetylase mutated, in complex with acetate [Bacillus subtilis],1ODT_H cephalosporin C deacetylase mutated, in complex with acetate [Bacillus subtilis] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| D5EXI2 | 9.31e-81 | 2 | 425 | 12 | 438 | Acetyl esterase Axe7A OS=Prevotella ruminicola (strain ATCC 19189 / JCM 8958 / 23) OX=264731 GN=axe7A PE=1 SV=1 |
| P94388 | 6.15e-24 | 125 | 407 | 9 | 298 | Cephalosporin-C deacetylase OS=Bacillus subtilis (strain 168) OX=224308 GN=cah PE=1 SV=1 |
| Q9WXT2 | 6.20e-23 | 122 | 407 | 14 | 303 | Cephalosporin-C deacetylase OS=Thermotoga maritima (strain ATCC 43589 / DSM 3109 / JCM 10099 / NBRC 100826 / MSB8) OX=243274 GN=axeA PE=1 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as SP
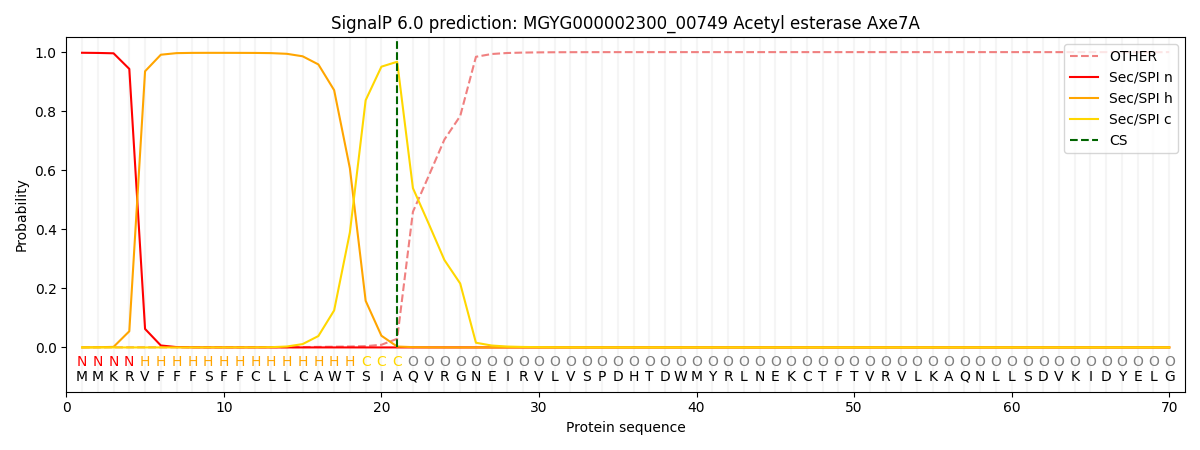
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.001542 | 0.995009 | 0.002713 | 0.000272 | 0.000226 | 0.000230 |
