You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000003419_00103
You are here: Home > Sequence: MGYG000003419_00103
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | RF16 sp900766775 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Bacteroidota; Bacteroidia; Bacteroidales; Paludibacteraceae; RF16; RF16 sp900766775 | |||||||||||
| CAZyme ID | MGYG000003419_00103 | |||||||||||
| CAZy Family | PL1 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 4010; End: 6322 Strand: - | |||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| PL1 | 220 | 390 | 6.4e-45 | 0.8366336633663366 |
| CBM77 | 588 | 688 | 5.7e-22 | 0.9611650485436893 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| COG3866 | PelB | 2.31e-57 | 183 | 507 | 68 | 342 | Pectate lyase [Carbohydrate transport and metabolism]. |
| pfam18283 | CBM77 | 1.63e-37 | 583 | 690 | 1 | 108 | Carbohydrate binding module 77. This domain is the non-catalytic carbohydrate binding module 77 (CBM77) present in Ruminococcus flavefaciens. CBMs fulfil a critical targeting function in plant cell wall depolymerisation. In CBM77, a cluster of conserved basic residues (Lys1092, Lys1107 and Lys1162) confer calcium-independent recognition of homogalacturonan. |
| smart00656 | Amb_all | 6.16e-34 | 227 | 392 | 17 | 190 | Amb_all domain. |
| pfam00544 | Pec_lyase_C | 9.39e-19 | 191 | 388 | 1 | 211 | Pectate lyase. This enzyme forms a right handed beta helix structure. Pectate lyase is an enzyme involved in the maceration and soft rotting of plant tissue. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| QUT73893.1 | 6.59e-235 | 12 | 724 | 1 | 716 |
| QNT67442.1 | 1.50e-196 | 33 | 692 | 108 | 773 |
| ATA84997.1 | 2.93e-187 | 33 | 540 | 40 | 541 |
| QOR20273.1 | 3.31e-186 | 33 | 540 | 50 | 551 |
| CBW15186.1 | 5.51e-186 | 33 | 540 | 65 | 566 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 3VMV_A | 3.75e-29 | 132 | 423 | 2 | 266 | Crystalstructure of pectate lyase Bsp165PelA from Bacillus sp. N165 [Bacillus sp. N16-5],3VMW_A Crystal structure of pectate lyase Bsp165PelA from Bacillus sp. N165 in complex with trigalacturonate [Bacillus sp. N16-5] |
| 1VBL_A | 1.93e-21 | 227 | 388 | 133 | 330 | Structureof the thermostable pectate lyase PL 47 [Bacillus sp. TS-47] |
| 2QXZ_A | 1.07e-18 | 212 | 404 | 63 | 261 | ChainA, pectate lyase II [Xanthomonas campestris pv. campestris],2QXZ_B Chain B, pectate lyase II [Xanthomonas campestris pv. campestris] |
| 2QY1_A | 1.07e-18 | 212 | 404 | 63 | 261 | ChainA, Pectate lyase II [Xanthomonas campestris pv. campestris],2QY1_B Chain B, Pectate lyase II [Xanthomonas campestris pv. campestris] |
| 2QX3_A | 2.60e-18 | 212 | 404 | 63 | 261 | Structureof pectate lyase II from Xanthomonas campestris pv. campestris str. ATCC 33913 [Xanthomonas campestris pv. campestris],2QX3_B Structure of pectate lyase II from Xanthomonas campestris pv. campestris str. ATCC 33913 [Xanthomonas campestris pv. campestris] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| Q65DC2 | 6.81e-28 | 174 | 397 | 67 | 281 | Pectate trisaccharide-lyase OS=Bacillus licheniformis (strain ATCC 14580 / DSM 13 / JCM 2505 / CCUG 7422 / NBRC 12200 / NCIMB 9375 / NCTC 10341 / NRRL NRS-1264 / Gibson 46) OX=279010 GN=BLi04129 PE=3 SV=1 |
| Q8GCB2 | 6.81e-28 | 174 | 397 | 67 | 281 | Pectate trisaccharide-lyase OS=Bacillus licheniformis OX=1402 GN=pelA PE=1 SV=1 |
| B1B6T1 | 6.81e-28 | 174 | 397 | 67 | 281 | Pectate trisaccharide-lyase OS=Bacillus sp. OX=1409 GN=pel PE=1 SV=1 |
| P0C1C2 | 1.85e-22 | 144 | 403 | 38 | 293 | Pectate lyase 3 OS=Pectobacterium carotovorum OX=554 GN=pel3 PE=1 SV=1 |
| P0C1C3 | 3.35e-22 | 156 | 403 | 25 | 293 | Pectate lyase 3 OS=Pectobacterium carotovorum subsp. carotovorum OX=555 GN=pel3 PE=3 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as SP
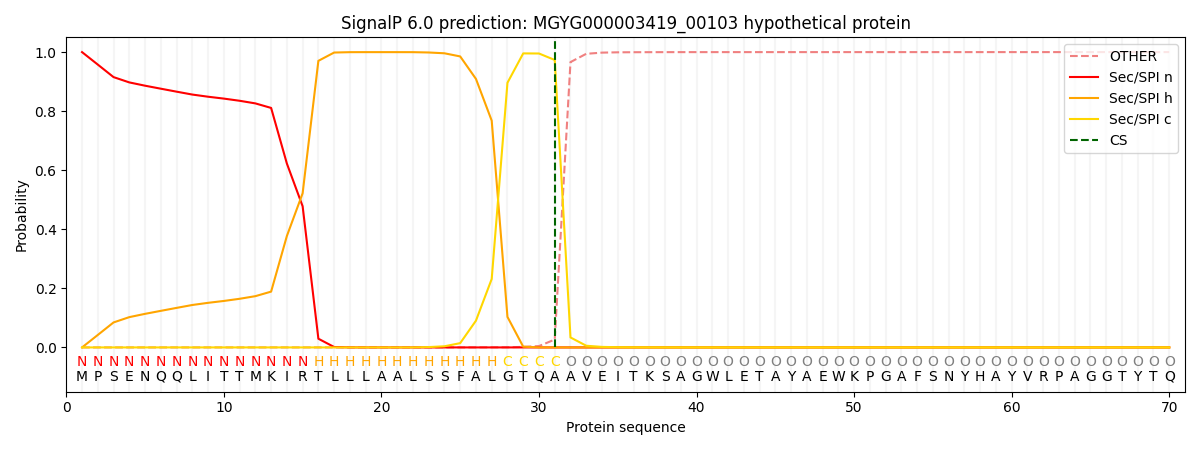
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.000259 | 0.999082 | 0.000173 | 0.000178 | 0.000154 | 0.000137 |
