You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000004090_02258
You are here: Home > Sequence: MGYG000004090_02258
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | CAG-279 sp900550025 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Bacteroidota; Bacteroidia; Bacteroidales; Muribaculaceae; CAG-279; CAG-279 sp900550025 | |||||||||||
| CAZyme ID | MGYG000004090_02258 | |||||||||||
| CAZy Family | GH29 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 4802; End: 6139 Strand: + | |||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| GH29 | 36 | 361 | 3.3e-100 | 0.9046242774566474 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| pfam01120 | Alpha_L_fucos | 1.59e-135 | 30 | 357 | 2 | 333 | Alpha-L-fucosidase. |
| smart00812 | Alpha_L_fucos | 2.71e-109 | 36 | 388 | 12 | 372 | Alpha-L-fucosidase. O-Glycosyl hydrolases (EC 3.2.1.-) are a widespread group of enzymes that hydrolyse the glycosidic bond between two or more carbohydrates, or between a carbohydrate and a non-carbohydrate moiety. A classification system for glycosyl hydrolases, based on sequence similarity, has led to the definition of 85 different families. This classification is available on the CAZy (CArbohydrate-Active EnZymes) web site. Because the fold of proteins is better conserved than their sequences, some of the families can be grouped in 'clans'. Family 29 encompasses alpha-L-fucosidases, which is a lysosomal enzyme responsible for hydrolyzing the alpha-1,6-linked fucose joined to the reducing-end N-acetylglucosamine of the carbohydrate moieties of glycoproteins. Deficiency of alpha-L-fucosidase results in the lysosomal storage disease fucosidosis. |
| COG3669 | AfuC | 1.09e-38 | 57 | 378 | 1 | 331 | Alpha-L-fucosidase [Carbohydrate transport and metabolism]. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| BBL00587.1 | 6.96e-262 | 34 | 443 | 5 | 413 |
| AFL78142.1 | 1.52e-261 | 34 | 443 | 26 | 434 |
| BBL08630.1 | 5.70e-261 | 34 | 443 | 5 | 413 |
| BBL11422.1 | 1.25e-260 | 34 | 443 | 26 | 434 |
| CBK63395.1 | 1.15e-258 | 32 | 443 | 23 | 433 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 6GN6_A | 2.52e-77 | 47 | 445 | 34 | 444 | Alpha-L-fucosidaseisoenzyme 1 from Paenibacillus thiaminolyticus [Paenibacillus thiaminolyticus],6GN6_B Alpha-L-fucosidase isoenzyme 1 from Paenibacillus thiaminolyticus [Paenibacillus thiaminolyticus],6GN6_C Alpha-L-fucosidase isoenzyme 1 from Paenibacillus thiaminolyticus [Paenibacillus thiaminolyticus],6GN6_D Alpha-L-fucosidase isoenzyme 1 from Paenibacillus thiaminolyticus [Paenibacillus thiaminolyticus],6GN6_E Alpha-L-fucosidase isoenzyme 1 from Paenibacillus thiaminolyticus [Paenibacillus thiaminolyticus],6GN6_F Alpha-L-fucosidase isoenzyme 1 from Paenibacillus thiaminolyticus [Paenibacillus thiaminolyticus] |
| 4PCS_A | 3.08e-48 | 43 | 413 | 18 | 405 | Crystalstructure of a bacterial fucosidase with iminosugar (2S,3S,4R,5S)-3,4-dihydroxy-2-[2'-phenyl]ethynyl-5-methylpyrrolidine [Bacteroides thetaiotaomicron VPI-5482],4PCS_B Crystal structure of a bacterial fucosidase with iminosugar (2S,3S,4R,5S)-3,4-dihydroxy-2-[2'-phenyl]ethynyl-5-methylpyrrolidine [Bacteroides thetaiotaomicron VPI-5482],4PCS_C Crystal structure of a bacterial fucosidase with iminosugar (2S,3S,4R,5S)-3,4-dihydroxy-2-[2'-phenyl]ethynyl-5-methylpyrrolidine [Bacteroides thetaiotaomicron VPI-5482],4PCS_D Crystal structure of a bacterial fucosidase with iminosugar (2S,3S,4R,5S)-3,4-dihydroxy-2-[2'-phenyl]ethynyl-5-methylpyrrolidine [Bacteroides thetaiotaomicron VPI-5482],4PCT_A Crystal structure of a bacterial fucosidase with iminocyclitol (2S,3S,4R,5S)-3,4-dihydroxy-2-ethynyl-5-methylpyrrolidine [Bacteroides thetaiotaomicron VPI-5482],4PCT_B Crystal structure of a bacterial fucosidase with iminocyclitol (2S,3S,4R,5S)-3,4-dihydroxy-2-ethynyl-5-methylpyrrolidine [Bacteroides thetaiotaomicron VPI-5482],4PCT_C Crystal structure of a bacterial fucosidase with iminocyclitol (2S,3S,4R,5S)-3,4-dihydroxy-2-ethynyl-5-methylpyrrolidine [Bacteroides thetaiotaomicron VPI-5482],4PCT_D Crystal structure of a bacterial fucosidase with iminocyclitol (2S,3S,4R,5S)-3,4-dihydroxy-2-ethynyl-5-methylpyrrolidine [Bacteroides thetaiotaomicron VPI-5482] |
| 2WVT_A | 3.34e-48 | 43 | 413 | 22 | 409 | Crystalstructure of an alpha-L-fucosidase GH29 from Bacteroides thetaiotaomicron in complex with a novel iminosugar fucosidase inhibitor [Bacteroides thetaiotaomicron VPI-5482],2WVT_B Crystal structure of an alpha-L-fucosidase GH29 from Bacteroides thetaiotaomicron in complex with a novel iminosugar fucosidase inhibitor [Bacteroides thetaiotaomicron VPI-5482],2WVU_A Crystal structure of a Michaelis complex of alpha-L-fucosidase GH29 from Bacteroides thetaiotaomicron with the synthetic substrate 4- nitrophenyl-alpha-L-fucose [Bacteroides thetaiotaomicron VPI-5482],2WVU_B Crystal structure of a Michaelis complex of alpha-L-fucosidase GH29 from Bacteroides thetaiotaomicron with the synthetic substrate 4- nitrophenyl-alpha-L-fucose [Bacteroides thetaiotaomicron VPI-5482],2WVU_C Crystal structure of a Michaelis complex of alpha-L-fucosidase GH29 from Bacteroides thetaiotaomicron with the synthetic substrate 4- nitrophenyl-alpha-L-fucose [Bacteroides thetaiotaomicron VPI-5482],2WVU_D Crystal structure of a Michaelis complex of alpha-L-fucosidase GH29 from Bacteroides thetaiotaomicron with the synthetic substrate 4- nitrophenyl-alpha-L-fucose [Bacteroides thetaiotaomicron VPI-5482] |
| 4WSJ_A | 3.48e-48 | 43 | 413 | 18 | 405 | Crystalstructure of a bacterial fucodiase in complex with 1-((1R,2R,3R,4R,5R,6R)-2,3,4-trihydroxy-5-methyl-7-azabicyclo[4.1.0]heptan-7-yl)ethan-1-one [Bacteroides thetaiotaomicron VPI-5482],4WSJ_B Crystal structure of a bacterial fucodiase in complex with 1-((1R,2R,3R,4R,5R,6R)-2,3,4-trihydroxy-5-methyl-7-azabicyclo[4.1.0]heptan-7-yl)ethan-1-one [Bacteroides thetaiotaomicron VPI-5482],4WSJ_C Crystal structure of a bacterial fucodiase in complex with 1-((1R,2R,3R,4R,5R,6R)-2,3,4-trihydroxy-5-methyl-7-azabicyclo[4.1.0]heptan-7-yl)ethan-1-one [Bacteroides thetaiotaomicron VPI-5482],4WSJ_D Crystal structure of a bacterial fucodiase in complex with 1-((1R,2R,3R,4R,5R,6R)-2,3,4-trihydroxy-5-methyl-7-azabicyclo[4.1.0]heptan-7-yl)ethan-1-one [Bacteroides thetaiotaomicron VPI-5482] |
| 5I5R_A | 3.55e-48 | 43 | 413 | 19 | 406 | Crystalstructure of a bacterial fucosidase with iminocyclitol (2S,3S,4R,5S)-3,4-dihydroxy-2-ethynyl-5-methylpyrrolidine [Bacteroides thetaiotaomicron VPI-5482],5I5R_B Crystal structure of a bacterial fucosidase with iminocyclitol (2S,3S,4R,5S)-3,4-dihydroxy-2-ethynyl-5-methylpyrrolidine [Bacteroides thetaiotaomicron VPI-5482],5I5R_C Crystal structure of a bacterial fucosidase with iminocyclitol (2S,3S,4R,5S)-3,4-dihydroxy-2-ethynyl-5-methylpyrrolidine [Bacteroides thetaiotaomicron VPI-5482],5I5R_D Crystal structure of a bacterial fucosidase with iminocyclitol (2S,3S,4R,5S)-3,4-dihydroxy-2-ethynyl-5-methylpyrrolidine [Bacteroides thetaiotaomicron VPI-5482] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| P10901 | 9.73e-43 | 47 | 428 | 37 | 427 | Alpha-L-fucosidase OS=Dictyostelium discoideum OX=44689 GN=alfA PE=3 SV=1 |
| Q6AYS4 | 5.37e-36 | 34 | 435 | 26 | 444 | Plasma alpha-L-fucosidase OS=Rattus norvegicus OX=10116 GN=Fuca2 PE=2 SV=1 |
| Q99KR8 | 7.17e-35 | 34 | 402 | 28 | 402 | Plasma alpha-L-fucosidase OS=Mus musculus OX=10090 GN=Fuca2 PE=1 SV=1 |
| P49713 | 1.32e-34 | 34 | 363 | 18 | 353 | Putative alpha-L-fucosidase OS=Caenorhabditis elegans OX=6239 GN=W03G11.3 PE=3 SV=2 |
| Q9BTY2 | 6.68e-33 | 34 | 435 | 34 | 452 | Plasma alpha-L-fucosidase OS=Homo sapiens OX=9606 GN=FUCA2 PE=1 SV=2 |
SignalP and Lipop Annotations help
This protein is predicted as SP
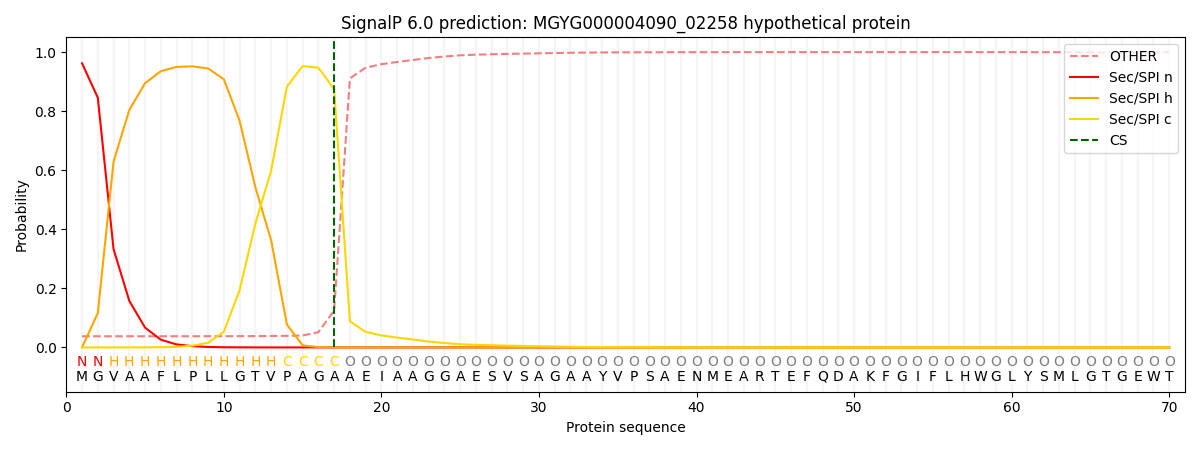
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.044506 | 0.953739 | 0.000369 | 0.000599 | 0.000390 | 0.000366 |
