You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000004691_01979
You are here: Home > Sequence: MGYG000004691_01979
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | UBA9502 sp900555625 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Firmicutes_A; Clostridia; Lachnospirales; Lachnospiraceae; UBA9502; UBA9502 sp900555625 | |||||||||||
| CAZyme ID | MGYG000004691_01979 | |||||||||||
| CAZy Family | GH0 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 448; End: 1785 Strand: + | |||||||||||
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| pfam01471 | PG_binding_1 | 4.62e-12 | 369 | 430 | 1 | 57 | Putative peptidoglycan binding domain. This domain is composed of three alpha helices. This domain is found at the N or C-terminus of a variety of enzymes involved in bacterial cell wall degradation. This domain may have a general peptidoglycan binding function. This family is found N-terminal to the catalytic domain of matrixins. The domain is found to bind peptidoglycan experimentally. |
| COG3409 | PGRP | 1.99e-07 | 364 | 430 | 39 | 102 | Peptidoglycan-binding (PGRP) domain of peptidoglycan hydrolases [Cell wall/membrane/envelope biogenesis]. |
| pfam13620 | CarboxypepD_reg | 1.96e-06 | 44 | 136 | 6 | 81 | Carboxypeptidase regulatory-like domain. |
| COG3409 | PGRP | 3.87e-06 | 360 | 432 | 117 | 185 | Peptidoglycan-binding (PGRP) domain of peptidoglycan hydrolases [Cell wall/membrane/envelope biogenesis]. |
| cd11308 | Peptidase_M14NE-CP-C_like | 0.003 | 44 | 132 | 6 | 71 | Peptidase associated domain: C-terminal domain of M14 N/E carboxypeptidase; putative folding, regulation, or interaction domain. This domain is found C-terminal to the M14 carboxypeptidase (CP) N/E subfamily containing zinc-binding enzymes that hydrolyze single C-terminal amino acids from polypeptide chains, and have a recognition site for the free C-terminal carboxyl group, which is a key determinant of specificity. The N/E subfamily includes enzymatically active members (carboxypeptidase N, E, M, D, and Z), as well as non-active members (carboxypeptidase-like protein 1, -2, aortic CP-like protein, and adipocyte enhancer binding protein-1) which lack the critical active site and substrate-binding residues considered necessary for activity. The active N/E enzymes fulfill a variety of cellular functions, including prohormone processing, regulation of peptide hormone activity, alteration of protein-protein or protein-cell interactions and transcriptional regulation. For M14 CPs, it has been suggested that this domain may assist in folding of the CP domain, regulate enzyme activity, or be involved in interactions with other proteins or with membranes; for carboxypeptidase M, it may interact with the bradykinin 1 receptor at the cell surface. This domain may also be found in other peptidase families. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| ASN94441.1 | 1.29e-227 | 29 | 443 | 3 | 415 |
| QRP40879.1 | 1.29e-227 | 29 | 443 | 3 | 415 |
| QJU21247.1 | 3.69e-227 | 29 | 443 | 3 | 415 |
| QIX92999.1 | 6.08e-226 | 29 | 443 | 3 | 415 |
| ANU48790.1 | 1.74e-225 | 29 | 443 | 3 | 415 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 1LBU_A | 3.99e-06 | 358 | 430 | 5 | 73 | HydrolaseMetallo (zn) Dd-peptidase [Streptomyces albus G] |
Swiss-Prot Hits help
SignalP and Lipop Annotations help
This protein is predicted as OTHER
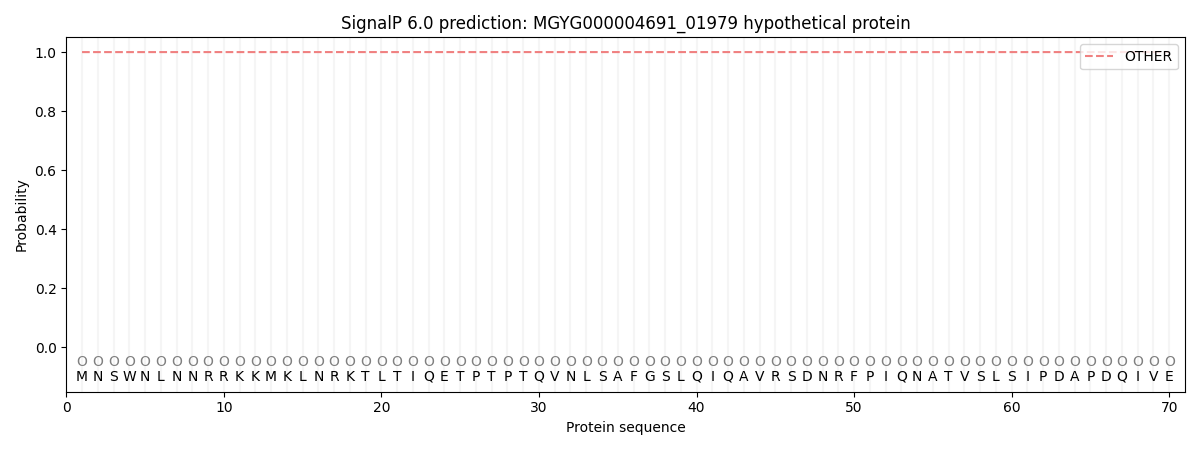
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 1.000046 | 0.000000 | 0.000000 | 0.000000 | 0.000000 | 0.000000 |
